Empowering
Cardiovascular Innovation
Through Computational Excellence
Diagnics Inc. delivers non-invasive, patient-specific computational biomechanics to transform cardiovascular diagnostics, prognostics, and intervention planning.
Where Science and Engineering Drive Clinical Impact
Leveraging proprietary technologies, Diagnics Inc. delivers noninvasive, rapid, and cost-effective heart analysis and quantification through advanced, patient-specific computational modeling.
Diagnics’ computational expertise & technology:
Provides diagnostic & prognostic insights
Supports preclinical studies
Supports clinical studies
Facilitates FDA clearance
Serves
Unlike AI-based approximations, our mechanistic, patient-specific models ensure unmatched reliability, precision, and interpretability—tailored to each patient’s unique physiology.
Diagnics’ approach enables comprehensive, high-fidelity evaluations supporting every stage of therapy advancement and cardiovascular device development from concept to clinical application.
Trusted insights
Tailored evaluations
Transformative outcomes
Why Diagnics’ Computational service?
Current diagnostic methods have restrictive limitations that hinder cardiac device innovations.
Diagnics’ computational technology bridges the gaps current clinical diagnostic methods.
❌ Clinical Diagnostic methods
✅ Diagnics’ Computational Solution
Cardiac Catheterization
Invasive, risky, and impractical for routine use.
Non-Invasive Modeling
Safe, repeatable, and patient-specific.
MRI
Poor temporal resolution, high cost, and not suitable for patients with implants.
High-Fidelity Simulations
Fast, cost-effective, and universally applicable.
Doppler Echocardiography
Lacks 3D hemodynamic and biomechanical detail.
3D Biomechanical Insight
Detailed, quantitative, and clinically actionable.
Furthermore …
Doppler-Based Ventricular Pressure-Volume (P-V) Loop Analysis
Our technology enables non-invasive P-V loop analysis using noninvasive Doppler echocardiography data.
P-V loop analysis can be used to quantify the impact of cardiac diseases, interventions, surgeries, or medications on cardiac function, hemodynamics, and left ventricle remodeling.
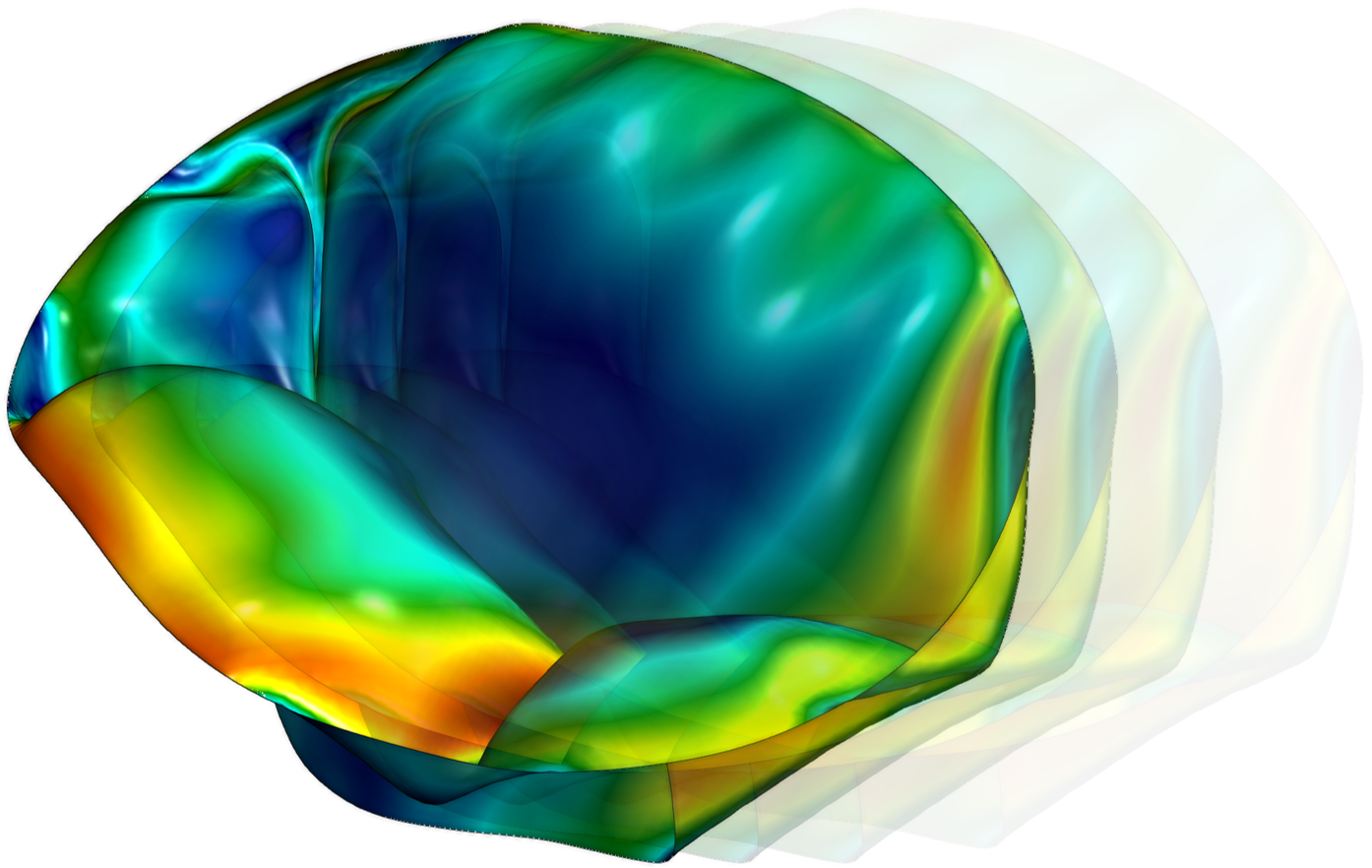
Analysis of Heart Valve Dynamics
This analysis quantifies the three-dimensional distribution of strain, stress, and leaflet-specific material properties in heart valves.
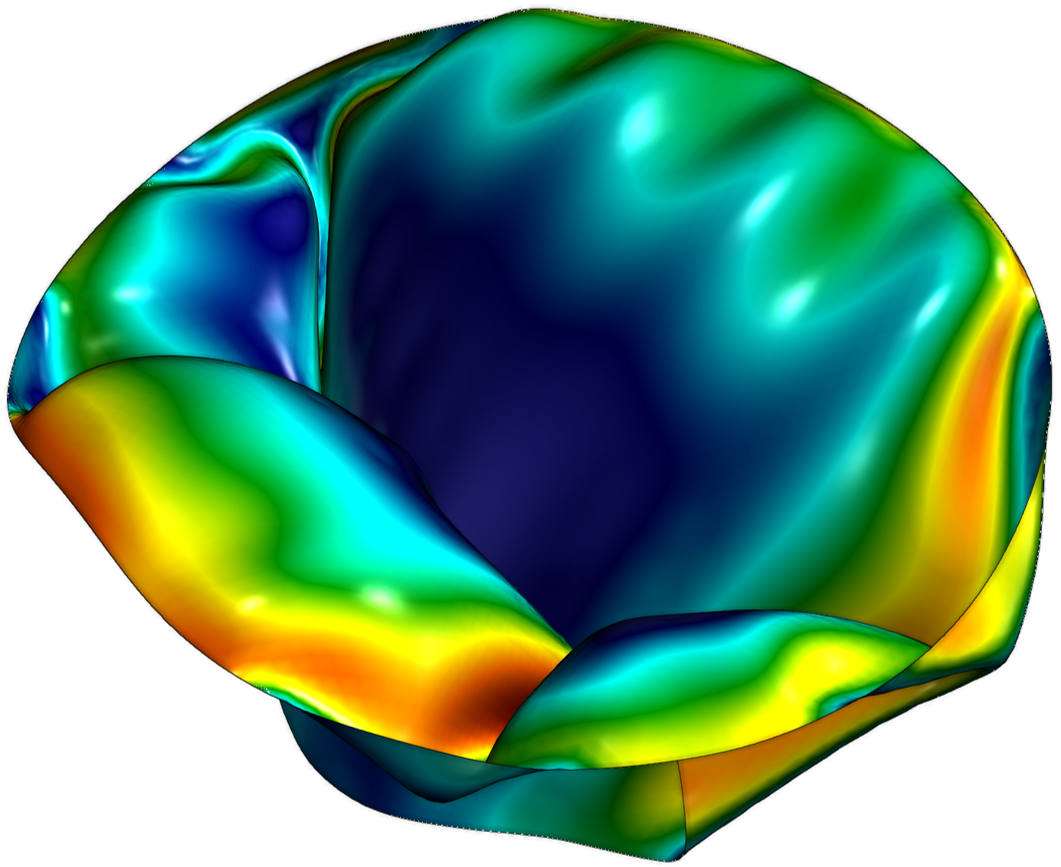
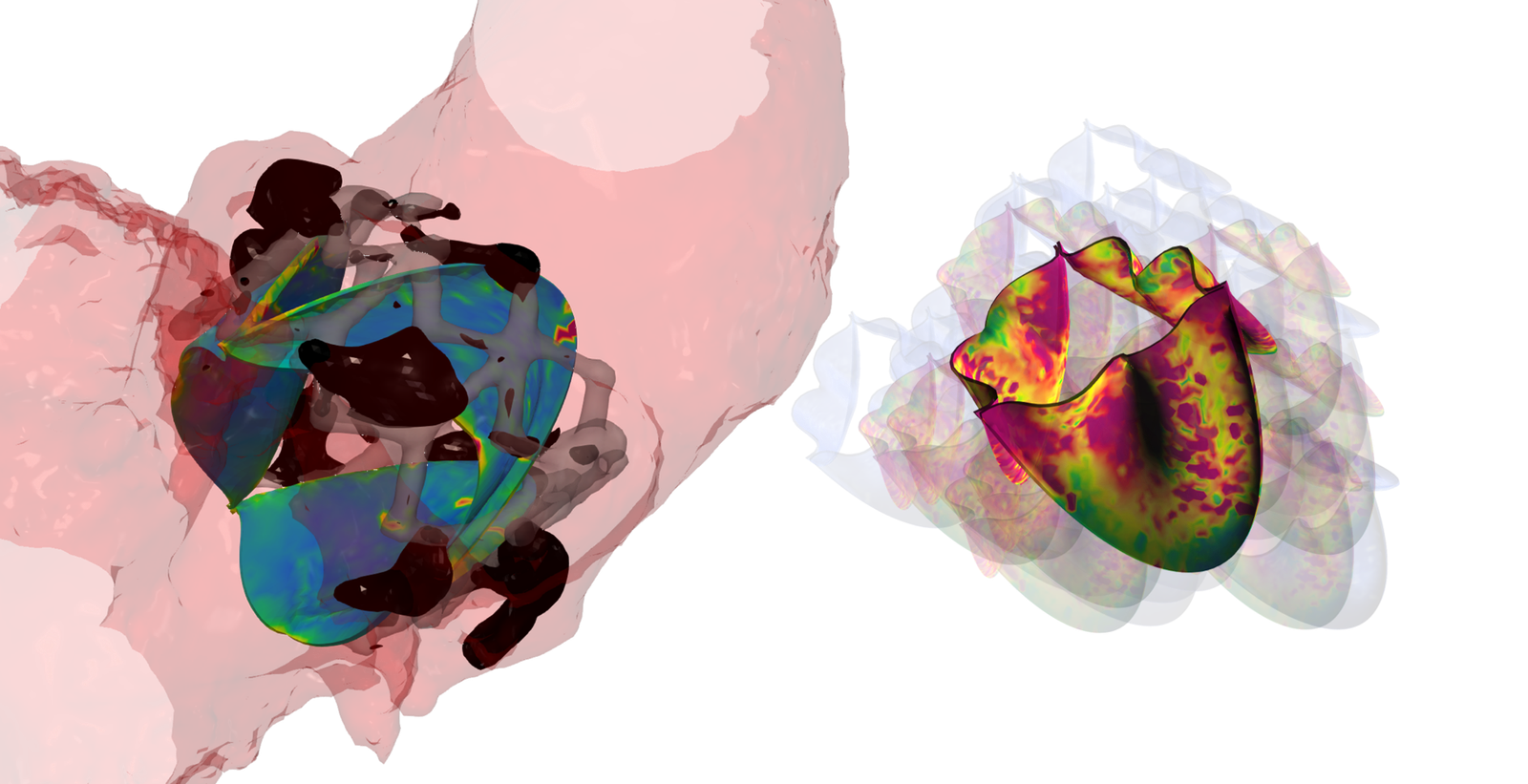
Mechanical stresses are a primary contributor to the failure of both native and prosthetic valves, serving as strong prognostic indicators of valvular disease progression and implant durability.
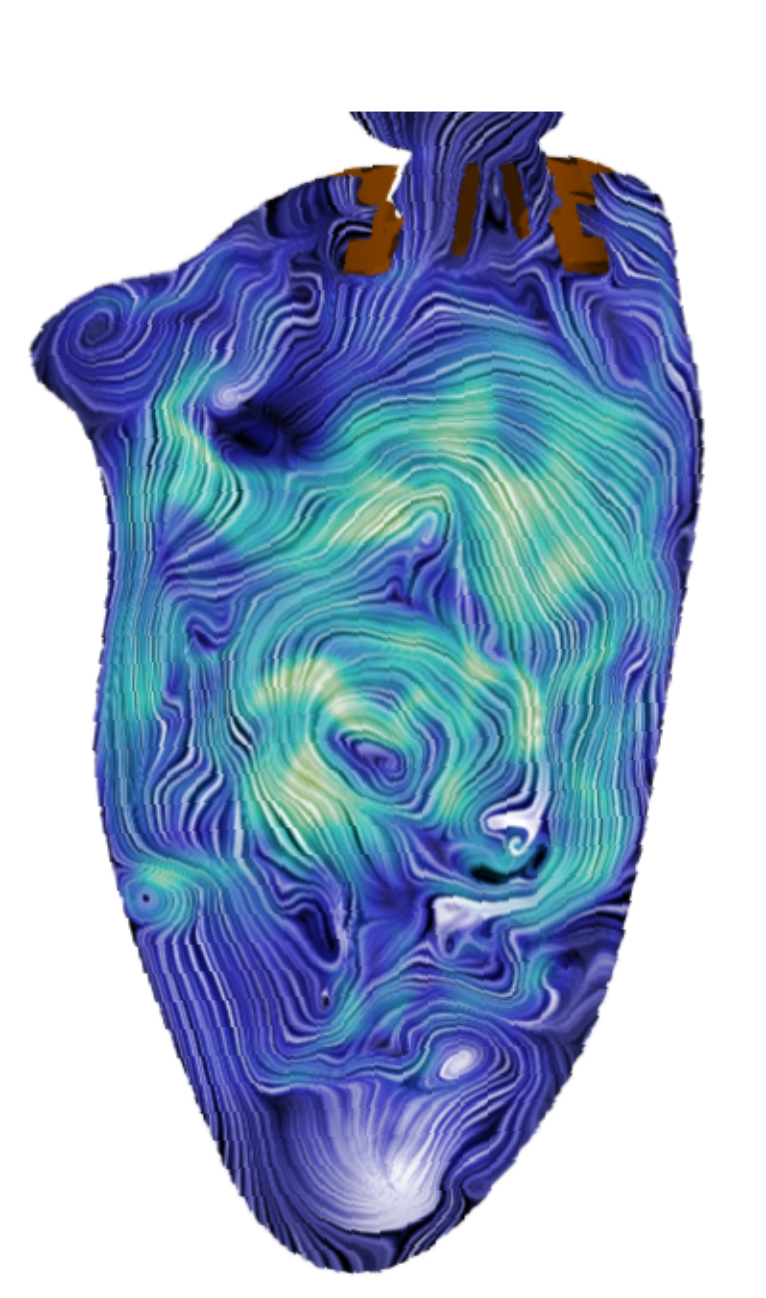
Patient-Specific Blood Flow Analysis
This analysis supports clinical teams, medical device innovators, and manufacturers in conducting detailed investigations of patient-specific blood flow dynamics.
By leveraging advanced computational modeling and simulation techniques, it enables precise evaluation of hemodynamic performance.
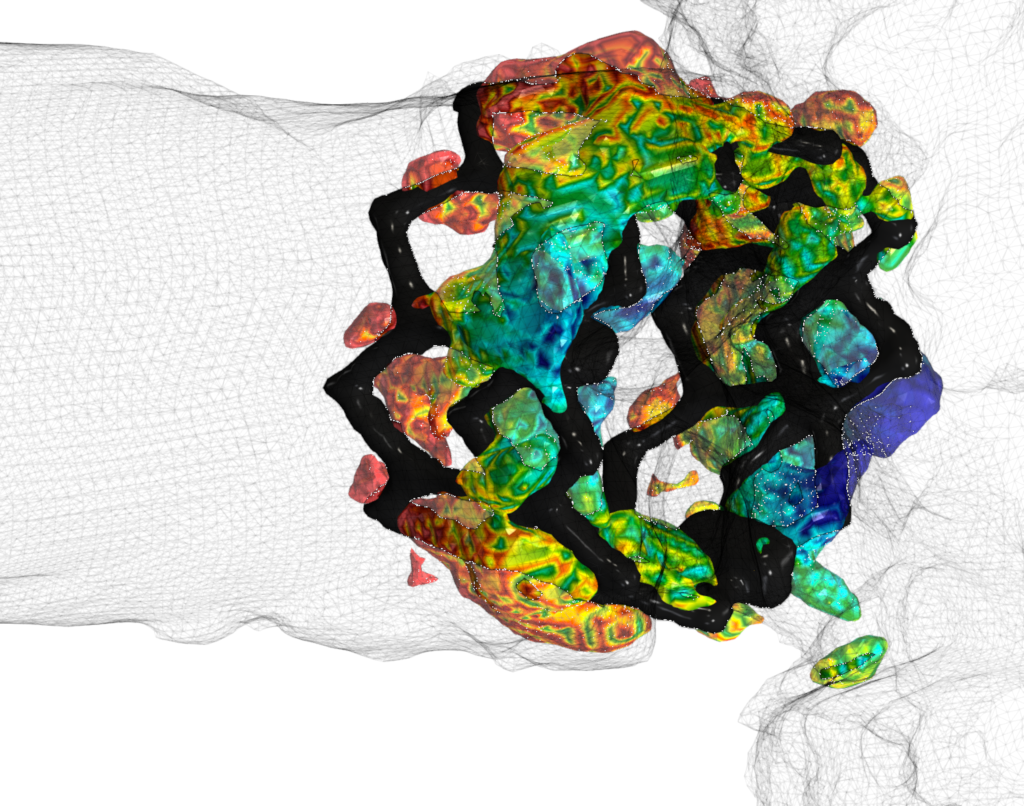
Vortex and micro – vortex analysis
(aortic valve)
These insights play a pivotal role in guiding the design, refinement, and rigorous validation of cardiovascular devices, directly contributing to their safety, effectiveness, and reliability in real-world clinical settings.
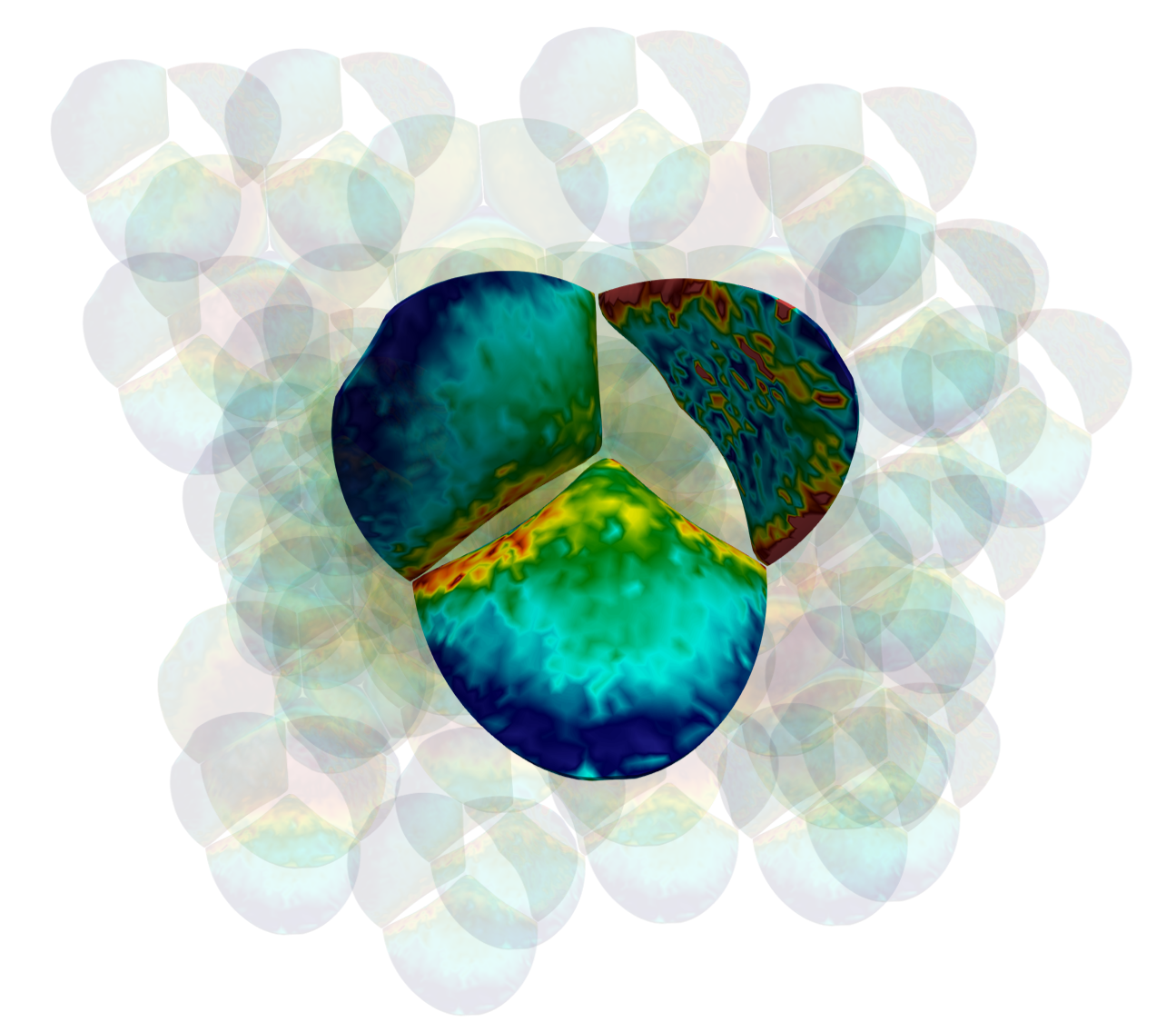
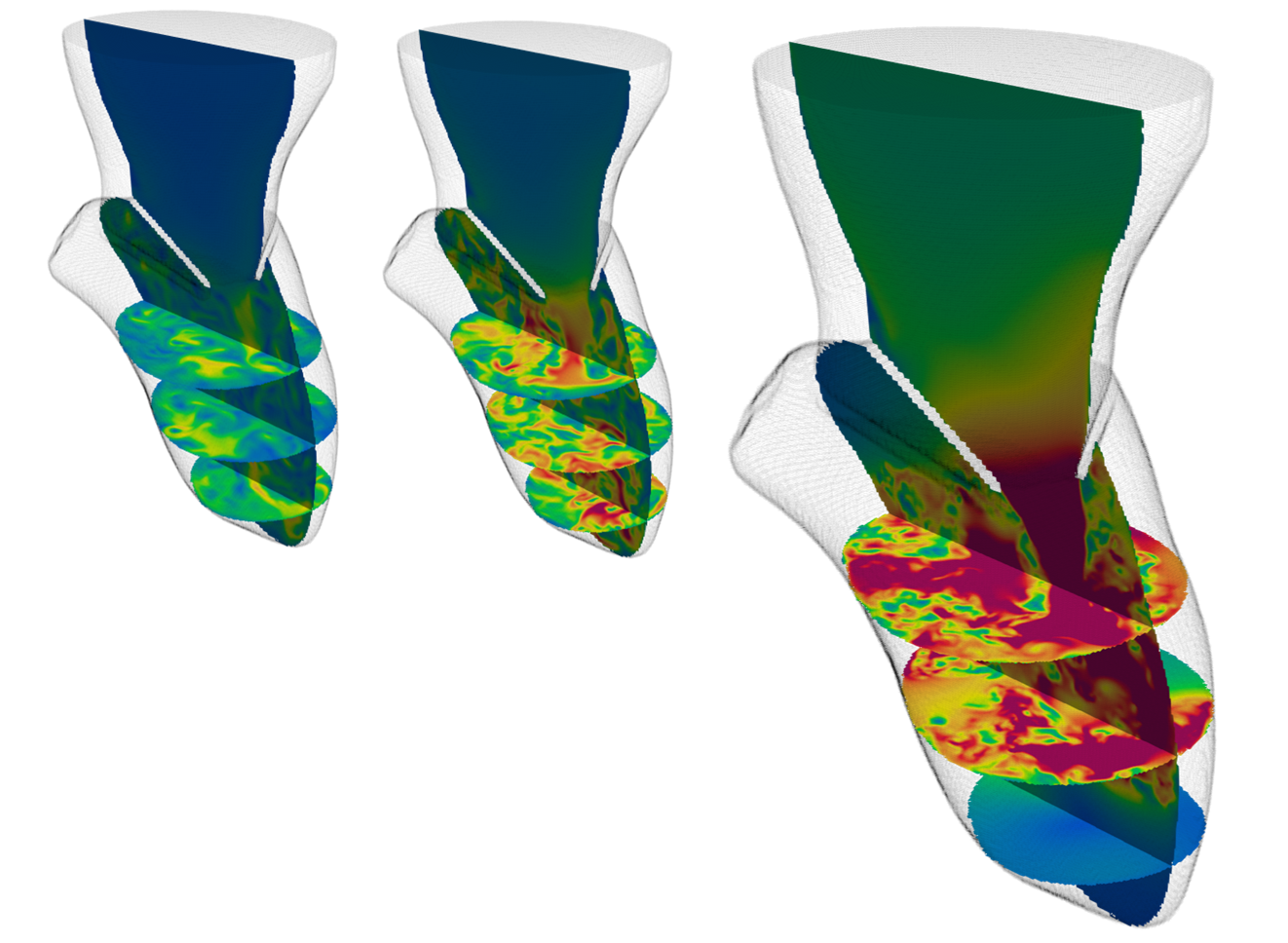
Intraventricular flow and vortex analysis post-TMVR
Intraventricular vortex analysis with prosthetic vs native mitral valves
Bileaflet mechanical mitral valve
Effective orifice area (cm2): 4.8
Mean pressure gradient (mmHg): 2.1
Vortex formation time: 6.4
Sphrecity index: 1.5
Trileaflet mitral bioprosthesis valve
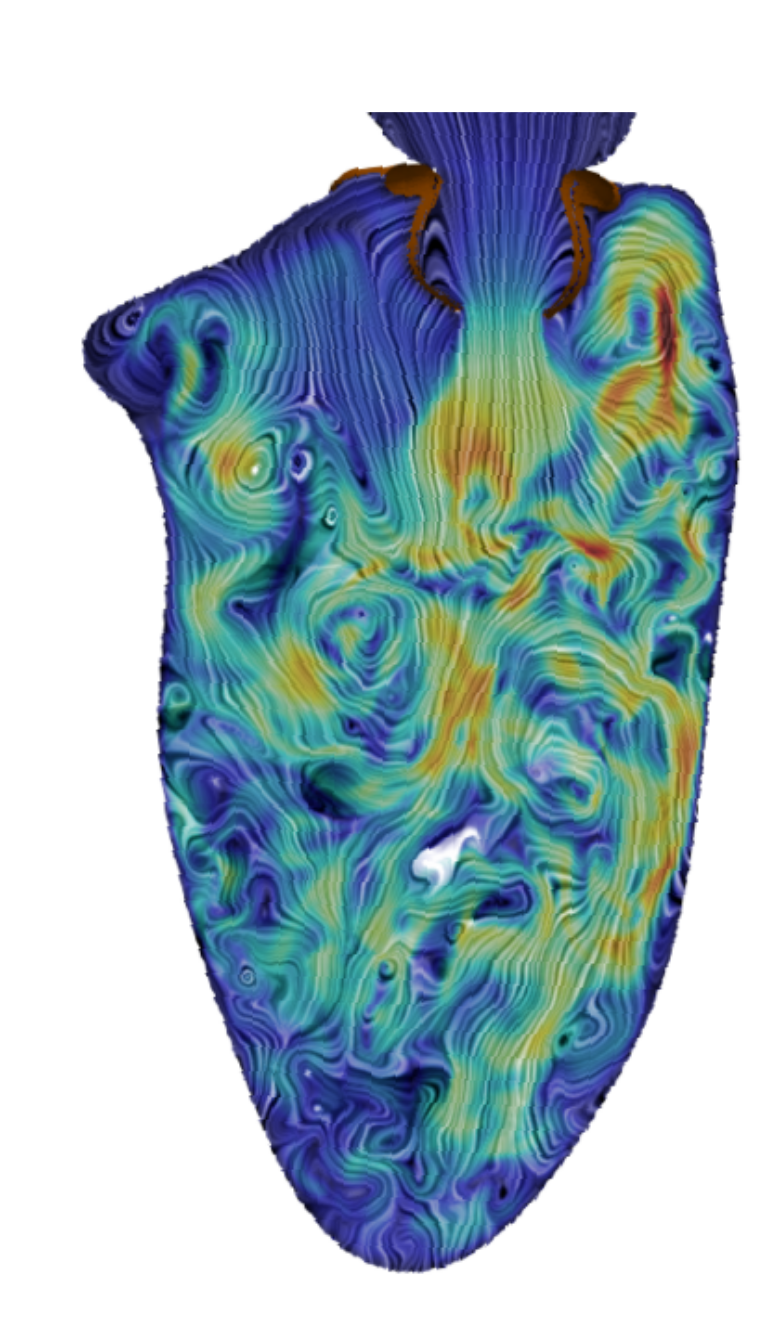

Effective orifice area (cm2): 3.3
Mean pressure gradient (mmHg): 2.4
Vortex formation time: 12.2
Sphrecity index: 1.2
Native mitral valve


Effective orifice area (cm2): 5.1
Mean pressure gradient (mmHg): 0.5
Vortex formation time: 3.5
Sphrecity index: 2.0