From clinical idea to
effective
cardiovascular devices
Preclinical CRO & Engineering Support for Every Stage of Cardiovascular Device Development
With over 20 years of experience, our team operates as a specialized preclinical CRO, transforming innovative concepts into life-saving technologies. From design to implementation, we provide comprehensive engineering and contract research services to ensure groundbreaking ideas become reality. Our approach helps innovators de-risk designs, accelerate development timelines, and navigate regulatory requirements with confidence.
Benefits of Partnering with DIAGNICS
Gain a Trusted Engineering Partner with 20+ Years of experience






Accelerate
Time-to-Market:
Bring your cardiovascular device from concept to clinical reality faster than ever. Our integrated approach combines advanced computational modeling, rapid prototyping, in vitro and ex vivo experimentation, and virtual trials to streamline every stage of development. By simulating performance under diverse physiological conditions and validating designs early, we minimize costly iterations and reduce regulatory hurdles. This means fewer delays, lower development costs, and a clear path to market, without compromising safety or efficacy.




De-Risk the Development Process:
Strengthen Regulatory Submissions:

At Diagnics, we recognize that a device’s long‑term performance is determined by its micro‑scale features. Every material choice, surface characteristic, and structural nuance is examined with scientific rigor to ensure nothing undermines safety or reliability.
Optimize Device Performance:
Accelerate Device Innovation with Virtual Testing
Cut development time and costs by simulating designs before building physical prototypes.
Predict Performance Across Diverse Patient Conditions
Ensure your device works safely and effectively for a wide range of anatomies and physiological scenarios.
Reduce Risk with Advanced Computational Modeling
Validate your design virtually to meet the highest standards of safety and efficacy.
Reduce Costly Prototypes and Animal Testing
Leverage simulation to minimize physical trials and streamline regulatory compliance.
Refine Designs Faster with Patient-Specific Analysis
Personalized modeling helps you optimize performance for real-world applications.
Achieve Confidence Before the First Prototype
Validate and iterate virtually to guarantee reliability and effectiveness from the start.
Connecting Breakthroughs to
Real-World Care
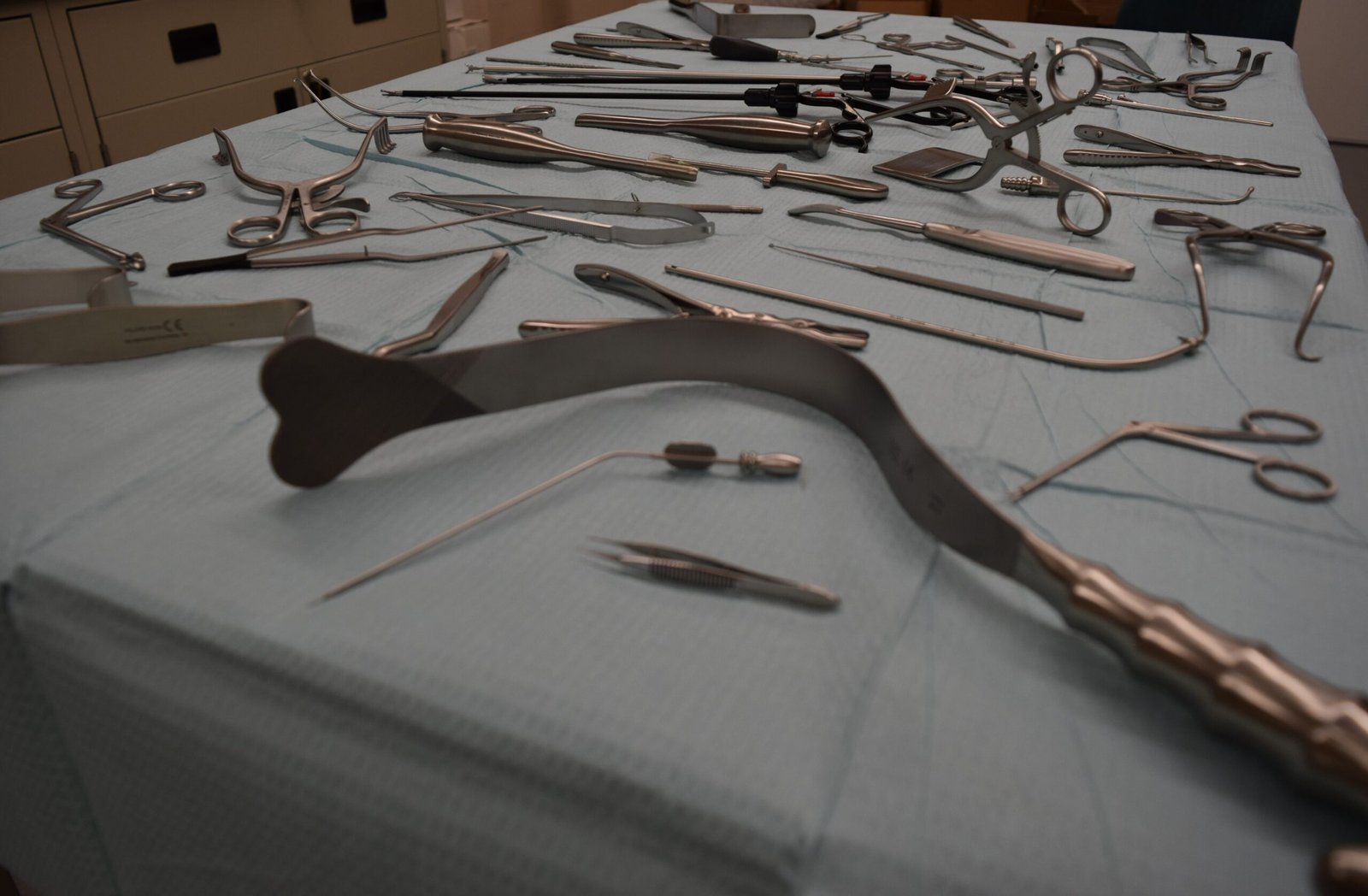
Seamlessly Bridge Research and Clinical Practice:
We believe a life-saving device must be be intuitive and reliable in the hands of the medical professionals who use it. Our design process seamlessly integrates with clinical feedback to optimize for usability, ergonomics, and seamless integration into surgical workflows. We ensure your device is not only technically superior but also practical, reducing surgical complexity and boosting confidence for the clinician. This holistic approach bridges the gap between the lab and the operating room, delivering a solution that is both highly functional and easy to deploy.
Do you have an innovative cardiovascular device idea?
Our engineering team—specialized in the design, research, and development of cardiovascular devices—offers comprehensive, end-to-end support to companies, clinical teams, and individual innovators. From conceptualization to bedside application, we guide you through every stage of development and commercialization, transforming your vision into a tangible, life-saving solution.